Randomized Controlled Trial of Physiology-Informed TMS (piTMS) for Major Depressive Disorder.
2026 Award: $199,991
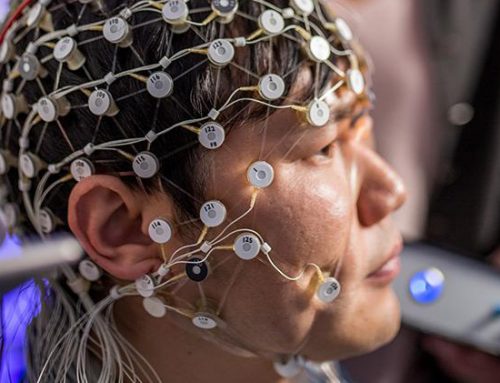
Current TMS treatments for depression use the same fixed timing pattern for every patient, even though real brain activity is irregular, fluctuating moment to moment. This study is testing a new personalized approach, physiology-informed TMS (piTMS), which uses a person’s own brain activity measured with EEG to build the timing of stimulation. By comparing piTMS directly against standard TMS in a randomized clinical trial, we aim to learn whether a more brain-derived stimulation approach can improve symptoms and produce stronger measurable changes in brain function.
Need/Problem: Major depressive disorder remains inadequately treated for many people, and even effective therapies can be limited by side effects, time burden, or incomplete remission. Although TMS is a well-established non-medication treatment, current protocols still rely on fixed, synthetic timing patterns that do not reflect the brain’s naturally irregular and individualized electrical activity, which may limit treatment response.
Grant Summary: Current TMS treatments for depression use fixed, synthetic timing patterns, even though the brain’s activity is highly irregular, dynamic, and individual. This mismatch may limit target engagement and contribute to variable treatment response. In this project, we will test physiology-informed TMS (piTMS), a new personalized TMS approach that derives each participant’s stimulation timing pattern from their own EEG, to determine whether better alignment with endogenous brain dynamics improves clinical and neurophysiological outcomes in major depressive disorder.
Goals & Projected Outcomes: The primary goals are to establish the safety, feasibility, and tolerability of accelerated piTMS and to estimate its preliminary clinical efficacy relative to standard iTBS in adults with depression. A key secondary goal is to test whether piTMS produces stronger target engagement, measured as a greater reduction in left prefrontal aperiodic exponent on HD-EEG, generating the pilot effect-size and mechanistic data needed for larger NIH-scale trials.

Flavio Frohlich, PhD

Athena Stein, PhD
Grant Details: This is a single-site, double-blind, randomized active-control pilot trial enrolling 28 adults with unipolar, non-psychotic major depressive disorder, randomized 1:1 to piTMS or standard iTBS. Both groups will complete a 2-day accelerated intervention consisting of five stimulation blocks per day, with each block delivering 1,800 pulses over 10 minutes at 90% resting motor threshold to the left dorsolateral prefrontal cortex; all participants will also receive adjunct d-cycloserine prior to daily stimulation to support neuroplasticity. High-density EEG and clinical assessments will be collected at baseline, end of intervention, and 2-week follow-up, allowing the study to assess both symptom change and whether individualized EEG-derived timing shifts the targeted broadband brain signal more effectively than standard TMS.